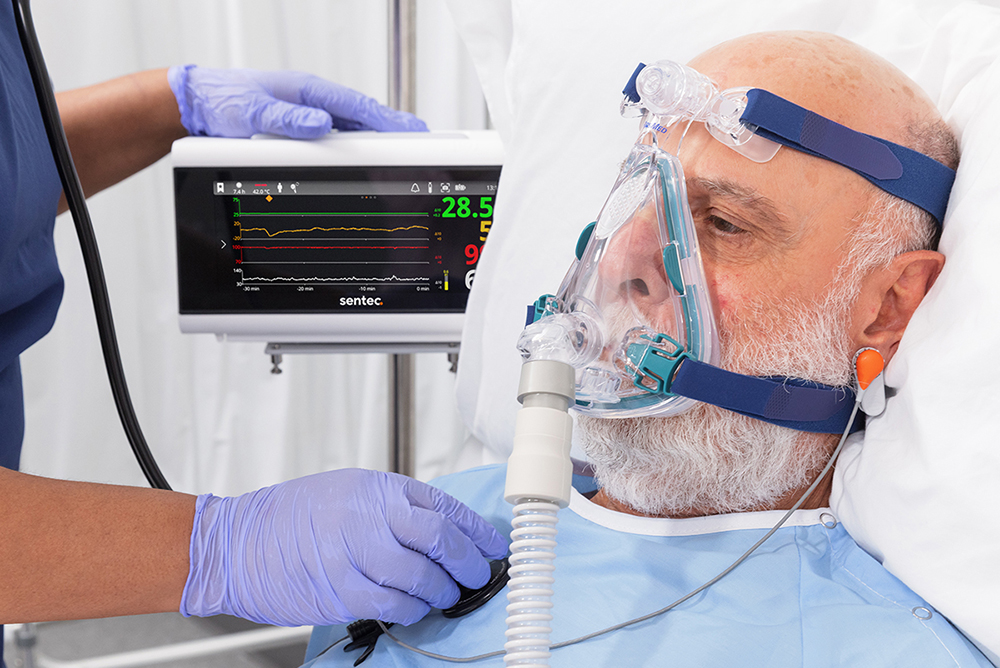
WHY TRANSCUTANEOUS CO2
Limitations of other CO2 Monitoring Methods
- Arterial blood gases (ABGs) are invasive and intermittent: While a reference standard, ABGs are invasive and offer only intermittent snapshots — potentially leaving variations in ventilation undetected.³
- SpO2 alone is insufficient: Measuring oxygenation (SpO2) alone is insufficient for detecting alveolar hypoventilation and can miss hypercapnia in patients on NIV.¹
- End-tidal (etCO2) can be unreliable: Ventilation–perfusion (V/Q) mismatch can lead to underestimation of arterial CO2 in populations such as COPD and obesity hypoventilation syndrome (OHS), while mask leaks and continuous flow during NIV further compromise measurement reliability.²
“Combining data from ventilator built‐in software and tcPCO2 seems to represent
the best strategy to detect poor NIV efficacy.”
BENEFITS FOR PATIENTS, PROVIDERS, AND FACILITIES
Greater Insights for More Informed Clinical Decision-Making
Transcutaneous CO2 monitoring delivers continuous, noninvasive assessment of ventilation via a sensor applied to the skin, offering greater accuracy than end-tidal CO2 and greater feasibility than ABGs. This technology can support informed clinical assessment and decision-making in sleep and NIV centers, including:
tCOM+
A System Designed for the Sleep Lab and NIV Management Units
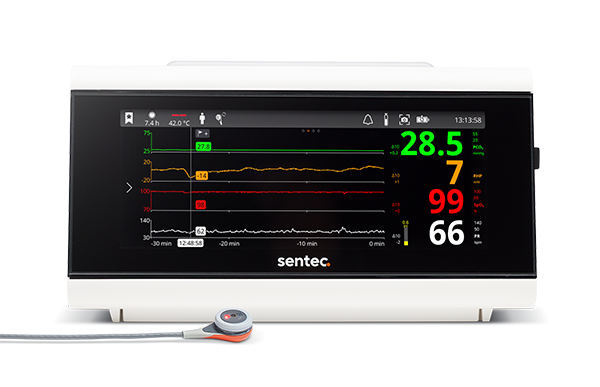
The tCOM+ Monitor features an intuitive, fully dimmable touchscreen display and silenceable alarms, and reports only stabilized values to PSG systems. In combination with the V-Sign™ Sensor, the system enables all-night monitoring without calibration or reapplication.
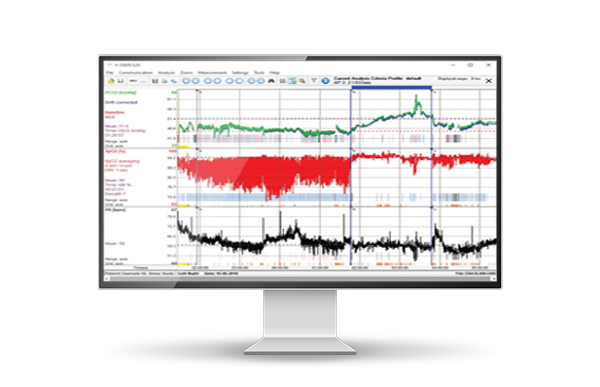
In combination with the included V-STATS Software, the system offers extensive functionalities to analyze and report measurement data (e.g. statistical analyses of CO2, delta values, and artifact detection) as well as broad connectivity to other devices, such as PSG systems.

Smart Cal-Mem feature allows temporary disconnection without recalibration, enabling hassle-free nighttime restroom visits.
Recommended in Clinical Practice Guidelines Worldwide
European Respiratory Society Guidelines on Long-Term Home Non-Invasive Ventilation for Management of COPD, 2019⁷
“Nocturnal monitoring of gas exchange is common practice in clinical settings. The use of PtcCO2 enables the detection of nocturnal hypoventilation, as modern devices show only minimal deviations.”
German Sleep Society, 2017⁶
“Transcutaneous capnometry is recommended as the most sensitive method for demonstrating the presence of sleep-related hypercapnia.”
American Academy of Sleep Medicine Manual for the Scoring of Sleep and Associated Events, 2018.⁴
“During PAP titration in both adults and children, arterial or transcutaneous PCO2 are the recommended methods to detect hypoventilation.”
American Association of Sleep Technologists Technical Guideline on Transcutaneous CO2 Monitoring, 2018⁵
“Within a sleep facility TCM is used to determine the presence of hypoventilation or respiratory depression during both diagnostic and treatment sleep testing, specifically as follows:
- During the collection of diagnostic polysomnography where there is a need for continuous monitoring of carbon dioxide levels during sleep.
- To assess a patient’s physiologic response to, and tailor treatment during therapeutic interventions (i.e., PAP therapy, including BiLevel, ASV and VAPS) during polysomnography.”
References
- Nardi, J., et al. Nocturnal Oximetry and Transcutaneous Carbon Dioxide in Home-Ventilated Neuromuscular Patients. Respir Care. 2012.
- Chhajed, P.N., et al. Utility of Transcutaneous Capnography for Optimization of Non-Invasive Ventilation Pressures. J Clin Diagn Res. 2016.
- Storre, J.H., et al. Monitoring of patients receiving mechanical ventilation. Pneumologie. 2014.
- Berry, R.B., et al. AASM Manual for the Scoring of Sleep and Associated Events: Rules, Terminology and Technical Specifications. 2018.
- American Association of Sleep Technologists Technical Guideline on Transcutaneous CO2 Monitoring. 2018.
- Mayer, G., et al. German S3 Guideline Nonrestorative Sleep/Sleep Disorders, chapter “Sleep-Related Breathing Disorders in Adults,” short version. Somnologie. 2017.
- Ergan, B., et al. European Respiratory Society guidelines on long-term home non-invasive ventilation for management of COPD. Eur Respir J. 2019.
Still have questions?
Complete this form for additional product support. After you’ve submitted your query, the appropriate Sentec associate will reach out with more information.